Difference between revisions of "Stable carbon isotopes in trees"
(Create Stable Carbon Isotopes In Trees) |
m (Fix link to D13C) |
||
| (2 intermediate revisions by one other user not shown) | |||
| Line 1: | Line 1: | ||
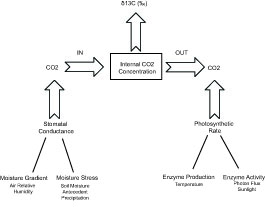
[[File:tree_13C_climatecontrol.jpg|thumb|alt=schematic of δ<sup>13<sup> controls in tree cellulose.|Fig. 1: Model of the influence of climate [[variable]]s on the carbon isotopic composition of plant cellulose <ref name="mccarroll2001">McCarroll, D. and F. Pawellek, Stable carbon isotope ratios of ''Pinus sylvestris'' from northern Finland and the potential for extracting a climate signal from long Fennoscandian chronologies. The Holocene, 2001. 11: p. 517-526.</ref>.]] | [[File:tree_13C_climatecontrol.jpg|thumb|alt=schematic of δ<sup>13<sup> controls in tree cellulose.|Fig. 1: Model of the influence of climate [[variable]]s on the carbon isotopic composition of plant cellulose <ref name="mccarroll2001">McCarroll, D. and F. Pawellek, Stable carbon isotope ratios of ''Pinus sylvestris'' from northern Finland and the potential for extracting a climate signal from long Fennoscandian chronologies. The Holocene, 2001. 11: p. 517-526.</ref>.]] | ||
| − | The source of carbon in trees is atmospheric [https://en.wikipedia.org/wiki/Carbon_dioxide CO<sub>2</sub>] The CO<sub>2</sub> enters the tree though the leaf stomata. An isotopic fractionation or discrimination against the [[ | + | The source of carbon in trees is atmospheric [https://en.wikipedia.org/wiki/Carbon_dioxide CO<sub>2</sub>] The CO<sub>2</sub> enters the tree though the leaf stomata. An isotopic fractionation or discrimination against the [[D13C | heavy isotope <sup>13</sup>C]] occurs during the diffusion through the [https://en.wikipedia.org/wiki/Stoma stomata]. Generally, the heavier isotopic species [https://en.wikipedia.org/wiki/Diffusion diffuse] more slowly than the lighter isotopic species. The diffusivity of <sup>13</sup>CO<sub>2</sub> in air has been calculated to be 4.4% less than that of <sup>12</sup>CO<sub>2</sub> <ref name="farquhar1982">Farquhar, G.D., M.H. O’Leary, and J.A. Berry, On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Australian Journal of Plant Physiology, 1982. 9: p. 121-137. </ref>. This simplistic value has been adopted to describe the fractionation due to diffusion through the leaf stomata. However, in reality, the situation is more complex since isotopes fractionation also occurs in the laminar boundary layer surrounding the leaf, during collision with guard cells, in the intracellular air spaces, and with water vapor <ref> Farquhar, G.D. and J. Llyod, Carbon and oxygen isotope effects in the exchange of carbon dioxide between terrestrial plants and the atmopshere, in Stable isotopes and plant carbon-water relations, J.R. Ehleringer, A.E. Hall, and G.D. Farquhar, Editors. 1993, Academic press: San Diego, CA. p. 47-70. </ref>. |
Another fractionation occurs during [https://en.wikipedia.org/wiki/Carboxylation carboxylation] by the enzyme used to fix carbon, which, for [https://en.wikipedia.org/wiki/C3_carbon_fixation C3 plants], is ribulose-1,5-biphosphate ([https://en.wikipedia.org/wiki/RuBisCO RuBisCO]). The magnitude of the fractionation depends on the [https://en.wikipedia.org/wiki/Photosynthesis photosynthetic] pathway (C3, C4, or CAM), the environment, the genotype and many other factors <ref> O’Leary, M.H., Biochemical basis of carbon isotope fractionation., in Stable isotopes and plant carbon-water relations, J.R. Ehleringer, A.E. Hall, and G.D. Farquhar, Editors. 1992, Academic Press: San Diego, CA. p. 19-28. </ref>. For C3 plants, this fractionation has been measured and is on the order of -27‰ <ref>Farquhar, G.D., et al., Effect of salinity and humidity on δ<sup>13</sup>C value of halophytes: evidence for diffusional isotope fractionation determined by the ratio of intercellular/ atmospheric partial pressure of CO2 under different environmental conditions. Oecologia, 1982. 52: p. 121-124. </ref>. | Another fractionation occurs during [https://en.wikipedia.org/wiki/Carboxylation carboxylation] by the enzyme used to fix carbon, which, for [https://en.wikipedia.org/wiki/C3_carbon_fixation C3 plants], is ribulose-1,5-biphosphate ([https://en.wikipedia.org/wiki/RuBisCO RuBisCO]). The magnitude of the fractionation depends on the [https://en.wikipedia.org/wiki/Photosynthesis photosynthetic] pathway (C3, C4, or CAM), the environment, the genotype and many other factors <ref> O’Leary, M.H., Biochemical basis of carbon isotope fractionation., in Stable isotopes and plant carbon-water relations, J.R. Ehleringer, A.E. Hall, and G.D. Farquhar, Editors. 1992, Academic Press: San Diego, CA. p. 19-28. </ref>. For C3 plants, this fractionation has been measured and is on the order of -27‰ <ref>Farquhar, G.D., et al., Effect of salinity and humidity on δ<sup>13</sup>C value of halophytes: evidence for diffusional isotope fractionation determined by the ratio of intercellular/ atmospheric partial pressure of CO2 under different environmental conditions. Oecologia, 1982. 52: p. 121-124. </ref>. | ||
Latest revision as of 17:53, 6 September 2016
The source of carbon in trees is atmospheric CO2 The CO2 enters the tree though the leaf stomata. An isotopic fractionation or discrimination against the heavy isotope 13C occurs during the diffusion through the stomata. Generally, the heavier isotopic species diffuse more slowly than the lighter isotopic species. The diffusivity of 13CO2 in air has been calculated to be 4.4% less than that of 12CO2 [2]. This simplistic value has been adopted to describe the fractionation due to diffusion through the leaf stomata. However, in reality, the situation is more complex since isotopes fractionation also occurs in the laminar boundary layer surrounding the leaf, during collision with guard cells, in the intracellular air spaces, and with water vapor [3].
Another fractionation occurs during carboxylation by the enzyme used to fix carbon, which, for C3 plants, is ribulose-1,5-biphosphate (RuBisCO). The magnitude of the fractionation depends on the photosynthetic pathway (C3, C4, or CAM), the environment, the genotype and many other factors [4]. For C3 plants, this fractionation has been measured and is on the order of -27‰ [5].
The δ13C of tree ring cellulose reflects the δ13C of internal CO2, whose isotopic composition is affected by the rate at which CO2 enters the leaf and the rate at which CO2 is taken up for photosynthesis, each step being associated with discrimination of 13CO2. The δ13C of cellulose of therefore a measure of the stomatal conductance, which is mainly controlled by moisture stress and humidity, and the photosynthetic rate, which depends on temperature and sunlight hours (Fig. 1) [1] [6] [7].
Quantitatively, carbon isotope fractionation in response to the atmospheric isotopic signature, diffusion through the stomata, carboxylation, and environmental factors can be described by the following equation [2]:
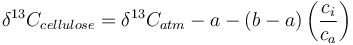
where:
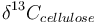 is the measured carbon isotopic composition of the cellulose
is the measured carbon isotopic composition of the cellulose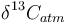 is the carbon isotopic signature of the atmosphere
is the carbon isotopic signature of the atmosphere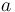 is the diffusion fractionation associated with the transport of CO2 though the stomata -4.4‰
is the diffusion fractionation associated with the transport of CO2 though the stomata -4.4‰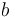 is the fractionation associated with carboxylation, -27‰
is the fractionation associated with carboxylation, -27‰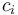 is the internal concentration of CO2, and
is the internal concentration of CO2, and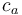 is the atmospheric carbon dioxide concentration.
is the atmospheric carbon dioxide concentration.
The 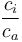 ratio is mainly controlled by environmental conditions. Over the past 150 years, the concentration of CO2 in the atmosphere has increased by more than 100 ppm. This carbon dioxide fertilization is thought to affect the tree physiology, and therefore, the δ13C of cellulose. Furthermore, this increase had been attributed to the anthropogenic burning of fossil fuels that are depleted in 13C relative to that of atmospheric CO2. Consequently, the carbon isotopic signature of the atmosphere has decreased over the last 150 years by about 1.5‰, a phenomenon known as the Seuss Effect]. This long-term trend is also reflected in plant δ13C. Corrections for the increased atmospheric CO2 and the change in the isotopic signature can be applied in order to retain only the climatic influence on the δ13C time series.
ratio is mainly controlled by environmental conditions. Over the past 150 years, the concentration of CO2 in the atmosphere has increased by more than 100 ppm. This carbon dioxide fertilization is thought to affect the tree physiology, and therefore, the δ13C of cellulose. Furthermore, this increase had been attributed to the anthropogenic burning of fossil fuels that are depleted in 13C relative to that of atmospheric CO2. Consequently, the carbon isotopic signature of the atmosphere has decreased over the last 150 years by about 1.5‰, a phenomenon known as the Seuss Effect]. This long-term trend is also reflected in plant δ13C. Corrections for the increased atmospheric CO2 and the change in the isotopic signature can be applied in order to retain only the climatic influence on the δ13C time series.
References
- ↑ 1.0 1.1 McCarroll, D. and F. Pawellek, Stable carbon isotope ratios of Pinus sylvestris from northern Finland and the potential for extracting a climate signal from long Fennoscandian chronologies. The Holocene, 2001. 11: p. 517-526.
- ↑ 2.0 2.1 Farquhar, G.D., M.H. O’Leary, and J.A. Berry, On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Australian Journal of Plant Physiology, 1982. 9: p. 121-137.
- ↑ Farquhar, G.D. and J. Llyod, Carbon and oxygen isotope effects in the exchange of carbon dioxide between terrestrial plants and the atmopshere, in Stable isotopes and plant carbon-water relations, J.R. Ehleringer, A.E. Hall, and G.D. Farquhar, Editors. 1993, Academic press: San Diego, CA. p. 47-70.
- ↑ O’Leary, M.H., Biochemical basis of carbon isotope fractionation., in Stable isotopes and plant carbon-water relations, J.R. Ehleringer, A.E. Hall, and G.D. Farquhar, Editors. 1992, Academic Press: San Diego, CA. p. 19-28.
- ↑ Farquhar, G.D., et al., Effect of salinity and humidity on δ13C value of halophytes: evidence for diffusional isotope fractionation determined by the ratio of intercellular/ atmospheric partial pressure of CO2 under different environmental conditions. Oecologia, 1982. 52: p. 121-124.
- ↑ McCarroll, D. and N.J. Loader, Stable isotopes in tree rings. Quaternary Science Reviews, 2004. 23(7-8): p. 771-801.
- ↑ McCarroll, D. and N.J. Loader, Isotopes in tree rings, in Isotopes in palaeoenvironmental research, M.J. Leng, Editor. 2006, Springer: The Netherlands. p. 67-116.